ABILITY
An implantable brain-computer interface (BCI) to restore movement and communication

Goal | restoring communication for paralyzed patients
Status | completed
Timeframe | 2018 – 2025
Area of Research | Bain-computer-interface technology
Partners | HUG, EPFL, UMC Utrecht, TU Graz, CHUV
Evolution | spun out as ABILITY Neurotech

Restoring communication
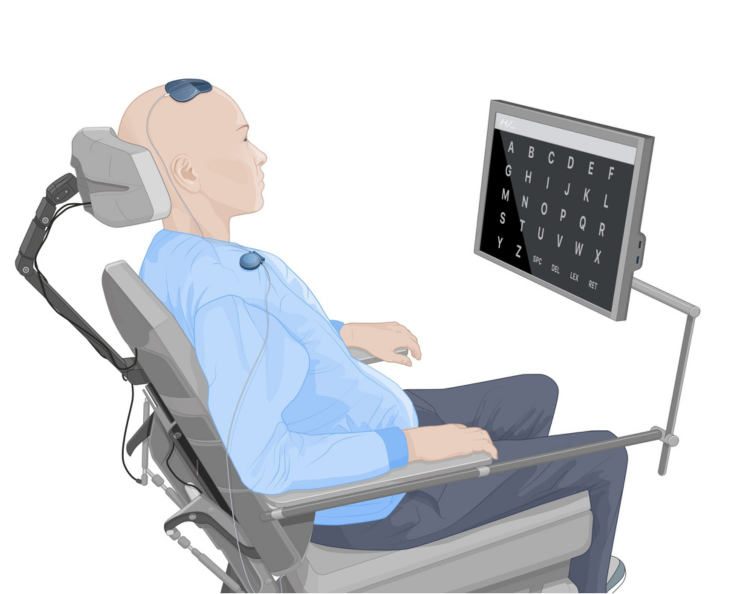
The Wyss Center is developing a fully implantable brain-computer interface (BCI) that can directly detect brain signals, and wirelessly transmit data through the scalp. The neural data is received by an external wearable device and then sent to a computer via a wired connection. The computer decodes the brain signals in real time to control assistive devices such as prosthetics or to integrate with voice or communication systems.
The system, called ABILITY which stands for Active Brain Implant Live Information Transfer sYstem, uses multiple channels to collect high-resolution brain data.
ABILITY in numbers
The ABILITY platform
The wearable is a component that comprises a head piece placed on the skin directly over the implant thanks to magnetic alignment, and a clip-piece. These wirelessly power the implant through the skin via induction.
The wearable also receives the neural data sent wirelessly by the implant and sends it to an external processing unit via a wired connection.
The external data processor uses the NeuroKey software to analyse the data, decode communication and movement intent, and send commands to computers or assistive devices in real time.
At the center of the ABILITY platform is an active, fully implantable medical device, designed for long-term implantation. Its design brings together state-of-the-art know-how and engineering to achieve ‘firsts’ in the area of brain implantable devices. With innovative features such as high density hermetic feedthroughs and optical data transmission, ABILITY can record brain signals from 128 channels at 30000 samples/s. Battery-less operation and encapsulation in a protective housing with hermetic sealing and biocompatible materials will allow the implant to function in the body for years.
The implant can be connected to microelectrodes arrays, subdural ECoG grids, or other kinds of electrodes. Hermetic feedthroughs allow the wires from the electrodes to enter and connect to the implant, while keeping moisture away from the sensitive internal electronics.
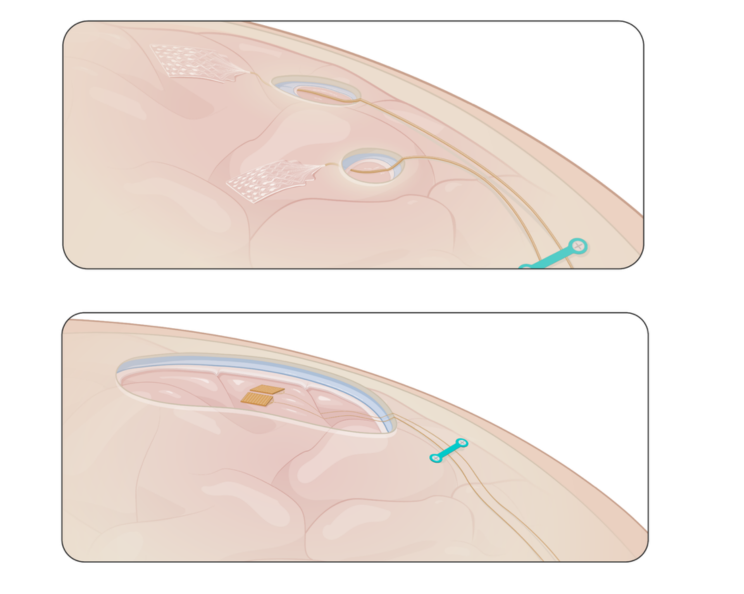
ABILITY’s preclinical trial
A preclinical trial has been performed with the ABILITY brain-computer interface system. The study, which has been carried out in sheep, is a crucial step towards development of a fully implantable device to enable applications such as communication and movement for people with paralysis. The trial has assessed the safety and feasibility of brain signal recording and wireless transfer of neural data to a wearable computer.
Image: top: subdural ECoG grids electrodes; bottom: Utah arrays electrodes

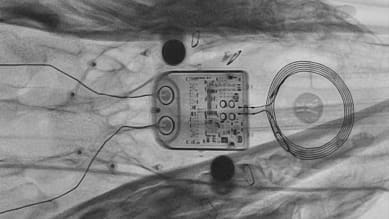
ABILITY comprises implantable and wearable components. Here, the image on the left shows the printed circuit board layout of the USB adapter – part of the ABILITY wearable. The image on the right is the physical board used in the wearable. Components marked yellow and blue in the layout are deep, so not visible on the board.
NeuroKey data processing software: The Wyss Center’s NeuroKey software is fully integrated with ABILITY. Developed as medical-grade software, the data analysis platform processes large amounts of information in real-time and enables rapid prototyping of clinical applications.




